You can Download Chapter 10 Haloalkanes and Haloarenes Questions and Answers, Notes, 2nd PUC Chemistry Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
Karnataka 2nd PUC Chemistry Question Bank Chapter 10 Haloalkanes and Haloarenes
2nd PUC Chemistry Haloalkanes and Haloarenes NCERT Textbook Questions
Question 1.
Name, the following halides according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
(i) (CH3)2CHCH(Cl)CH3
(ii) CH3CH2CH(CH3)CH(C2H5)Cl
(iii) CH3CH2C(CH3)2CH3I
(iv) (CH3)3CCH2CH(Br)C6H5
(v) CH3CH(CH3)CH(Br)CH3
(vi) CH3C(C2H5)2CH2Br
(vii) CH3C(Cl)(C2H5)CH2CH3
(viii) CH3CH=C(Cl)CH2CH(CH3)2
(ix) CH3CH=CHC(Br)(CH3)2
(x) P-ClC6H4CH2CH(CH3)2
(xi) m-CICH2C6H4CH2C(CH3)3
(xii) o-Br-C6H4CH(CH3)CH2CH3
Answer:
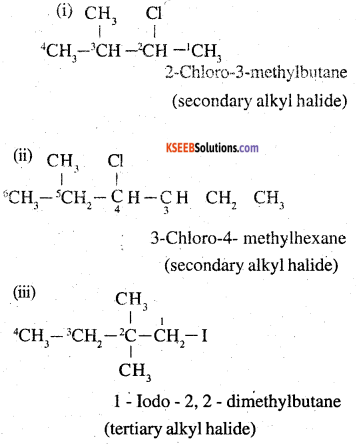
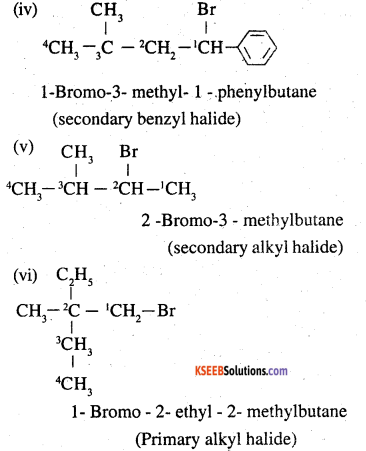
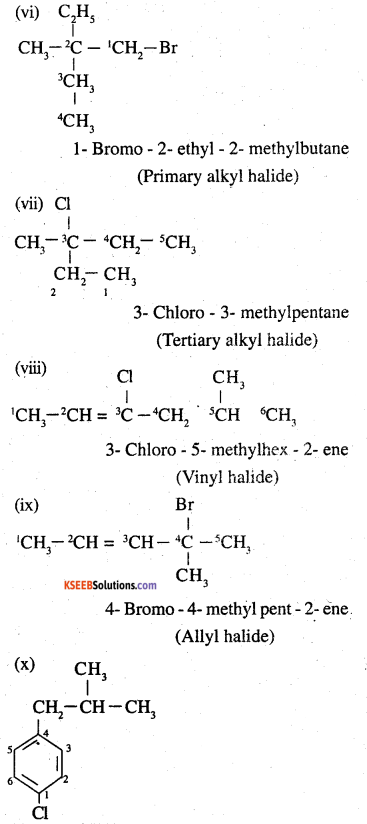
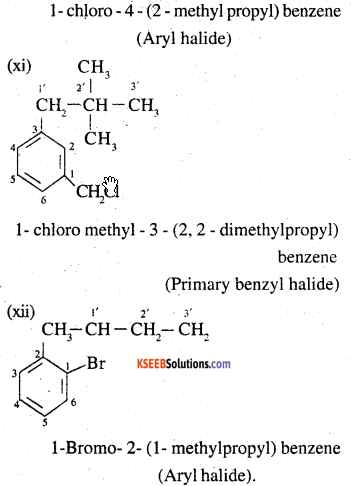
![]()
Question 2.
Give the IUPAC names of the following compounds:
(i) CH3CH(CI)CH(Br)CHCH3
(ii) CHF2CBrClF
(iii) ClCH2C = CCH2Br
(iv) (CCl3)3CCl
(v) CHCH3C(p-ClCCH6HCH4)CH2CH(Br)CHCH3
(vi) (CH3)3CCH=ClCC6`H4I-p
Answer:
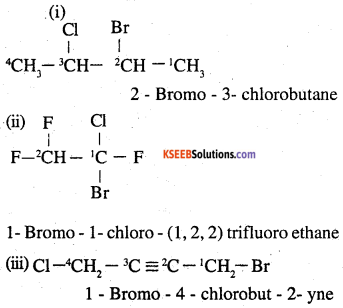
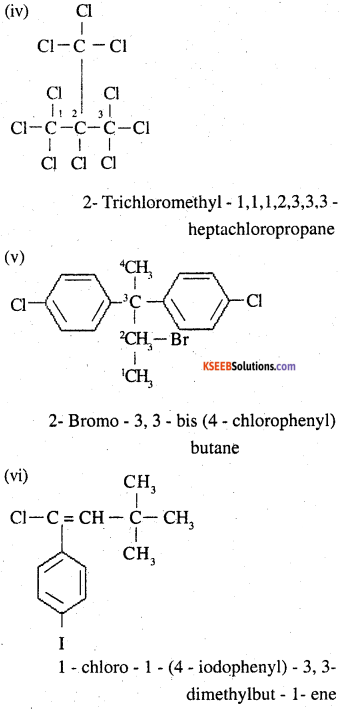
Question 3.
Write the structures of the following organic halogen compounds.
(i) 2-Chloro-3-methylpentane
(ii) p-Bromochlorobenzene
(iii) l-Chloro-4-ethylcyclohexane
(iv) 2-(2-Chlorophenyl)-l-iodooctane
(v) Perfluorobenzene
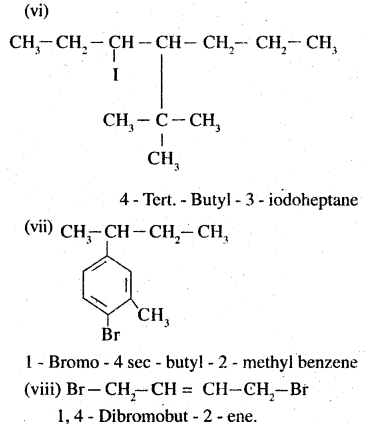
(vi) 4-tert-Butyl-3-iodoheptane
(vii) l-Bromo-4-sec-butyl-2-methyl- benzene .
(viii) l,4-Dibromobut-2-ene
Answer:
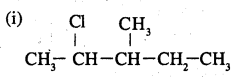
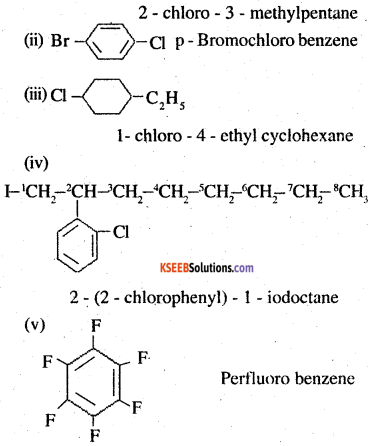
![]()
Question 4.
Which one of the following has the highest dipole moment?
(i) CH2Cl2
(ii) CHCl3
(iii) CCl4
Answer:
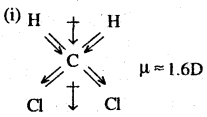
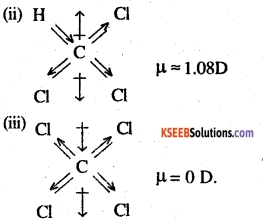
CCl4 is a symmetrical molecule. Therefore, the dipole moments of all four C-Cl bonds cancel each other. Hence its resultant dipole moment is zero.
As shown in the above figure, in CHCl3, the resultant dipole moments of two C-Cl bonds is opposed by the resultant dipole moments of one C-H and one C-Cl bond. Since the resultant of one C-H and one C-Cl bond is smaller than the resultant of the two C-Cl bonds dipole moments, the opposition is to a small extent. As a result CHCl3 has a small net dipole moment.
On the other hand, in case of CH2,Cl2 the resultant of the dipole moments of two C-Cl bonds is strengthened by the resultant of the dipole moments of two C-H bonds. As a result, CH2Cl2 has a higher dipole moment. Hence CH2Cl2 has the highest dipole moments among the three compounds.
Hence, the given compounds can be arranged in the increasing order of their dipole moments as
CCl4 < CHCl3 < CH2Cl2
Question 5.
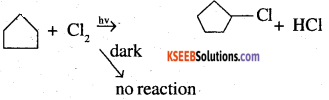
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Answer:
A hydrocarbon with molecular formula, C5H10 belongs to the group with a general molecular form CnH2n. therefore, it may either be an alkene or a cycloalkane since hydrocarbon does not react with chlorine in the dark, it cannot be alkene. Further, the hydrocarbon gives a single monochloro compound, C5H9Cl by reacting with chlorine in might sunshine since the formed compound is monochloro one all the C-H bonds should be equivalent. Hence the compound should be a cycloalkane. Hence the compound is C5H10 (cyclopentane).
Question 6.
Write the isomers of the compound having formula C4H9Br.
Answer:
There are four isomers of the compound having the formula C4H9Br. These isomers are given by

![]()
Question 7.
Write the equations for the preparation of 1-iodobutane from
(i) 1-butanol
(ii) 1-chlorobutane
(iii) but-l-ene.
Answer:
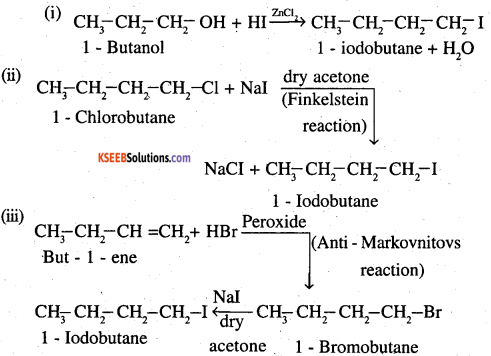
Question 8.
What are ambident nucleophiles? Explain with an example.
Answer:
Ambident nucleophiles are nucleophiles having two nucleophilic sites. Thus ambident nucleophiles have two sites through which they can attack. For example, nitrite ion is an ambident nucleophile.
![]()
Nitrite ion can attack through oxygen resulting in the formation of alkyl nitrites. Also, it can attack through nitrogen resulting in the formation of nitroalkanes.
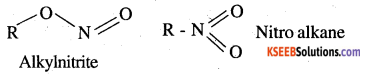
Question 9.
Which compound in each of the following pairs will react faster in SN2 reaction with OH?
1. CH3Br or CH3I
2. (CH3)3CCl or CH3Cl
Answer:
1. In the SN2 mechanism the reactivity of halides for the same alkyl group increase in the order. This happens because as the size increases the halide ion becomes a better leaving group.
R-F << R-Cl < R – Br < R-I
Therefore, CH3I will react faster than CH3Br in SN2 reaction with image 17.
2. The SN2 mechanism involves the attack of the nucleophile at the atom bearing the leaving group. But, in case (CH3)3 CCl, the attack of the nucleophile at the carbon atom is hindered by the presence of the bulky substituents on that carbon atom bearing-the leaving group in CH3Cl. Hence CH3Cl reacts faster than (CH3)3 CCl in SN2 reaction with ![]()
![]()
Question 10.
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene:
1. 1-Bromo-l-methylcyclohexane
2. 2-Chloro-2-methylbutane ,
3. 2,2,3-Trimethyl-3-bromopentane.
Answer:
1.
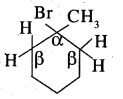
In this compound, all β hydrogens are equivalent thus dehydrohalogenation gives only one alkene.
2.
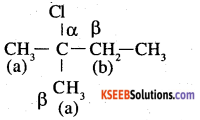
In the given compound, there are two different sets of equivalent P – hydrogen atoms labelled as ‘a’ and ‘b’ thus dehydrohalogenation yields two alkenes.
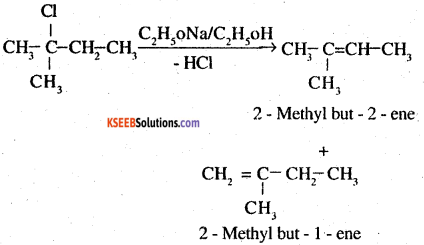
Sayt zeff’s rule implies that in dehydrohalogenate reactions, the alkene having a greater number of alkyl groups attached to a doubly bonded carbon atoms is preferably produced.
Therefore, 2-methyl but – 2- ene is the major product in the reaction.
3.
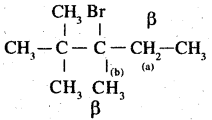
In the given compound, there are two different sets of equivalent P – hydrogen atoms labelled as ‘a’ and ‘b Thus dehydrogenation of the compound yields two alkenes.
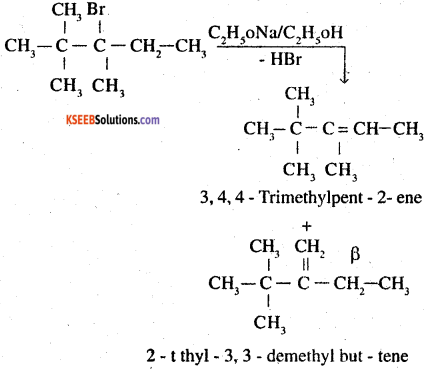
2 -1 thyl – 3, 3 – demethyl but – tene
Again according to Saytzeff’s rule
3,4,4 – trimethylpent – 2- ene is the major product.
Question 11.
How will you bring about the following conversions?
(i) Ethanol to but-l-yne
(ii) Ethane to bromoethene
(iii) Propene to 1-nitropropane
(iv) Toluene to benzyl alcohol
(v) Propene to propyne
(vi) Ethanol to ethyl fluoride
(vii) Bromomethane to propanone
(viii) But-l-ene to but-2-ene
(ix) 1-ChIorobutane to n-octane
(x) Benzene to biphenyl.
Answer:
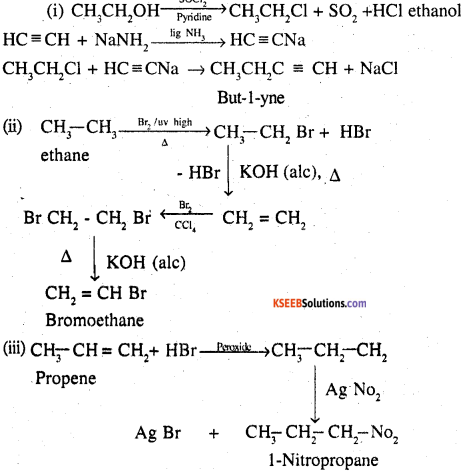
![]()
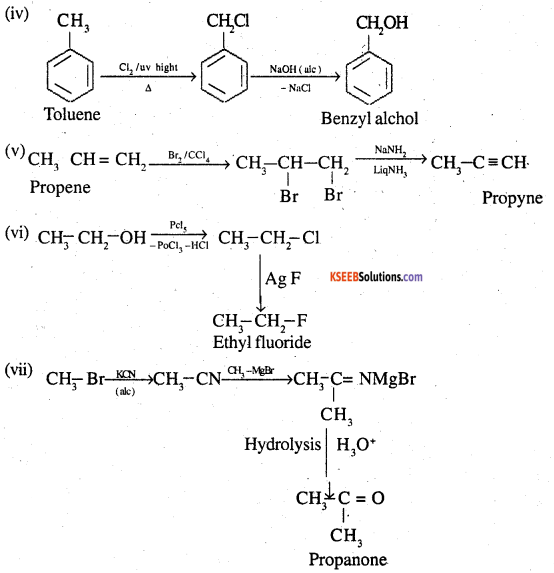
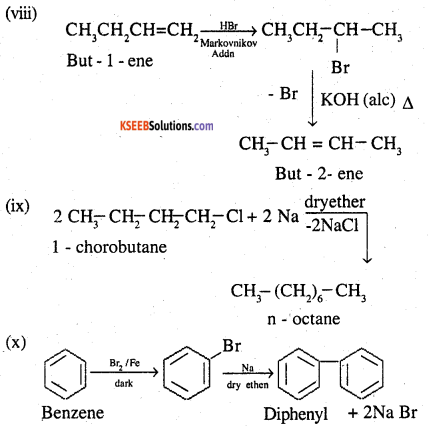
Question 12.
Explain why
1. The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride?
2. Alkyl halides, though polar, are immiscible with water?
3. Grignard reagents should be prepared under anhydrous conditions?
Answer:
1.
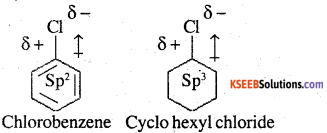
In chlorobenzene, the Cl-atom is linked to a sp2 hybridized carbon atom. In cyclohexyl chloride, the Cl atom is linked to a sp3 hybridized carbon. Now, sp2 hybridized carbon has more s-character than sp3 hybridized carbon atom. Therefore, the former is more electronegative than the latter. Therefore, the density of electrons of C-Cl bond near the Cl-atom is less in chlorobenzene than in cyclohexyl chloride. Moreover, the-R effect of the benzene ring of chlorobenzene decreases the electron density of the C-Cl bond near the Cl-atom. As a result, the polarity of C-Cl bond in chlorobenzene decreases. Hence, the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
2. To be miscible with water, the solute-water force of attraction must be stronger than the solute solute and water-water forces of attraction. Alkyl halides are polar molecules and so held together by dipole-dipole interactions. Similarly, strong H-bonds exist between the water molecules. The new force of attraction between the alkyl halides and water molecules is weaker than the alkyl-halide- alkyl halide and water-water forces of attraction. Hence, alkyl halides (though polar) are immiscible with water.
3. Grignard reagents are very reaction. In the presence of moisture, they react to give alkanes
R-Mg-X + H2O → RH + Mg(OH)X
Therefore, Grignard reagents should be prepared in anhydrous conditions.
Question 13.
Give the uses of freon 12, DDT, carbon tetrachloride and iodoform.
Answer:
Uses of freon – 12:
Freon -12 (dichlorodifluoromethane, CF2Cl2) is commonly known as CFC. It is used as a refrigerant in refrigerators and air conditioners. It is also used in aerosol spray propellants such as body sprays, hair sprays etc. However, it damages the ozone layer. Hence its manufacture is banned.
Uses of DDT:
DDT (p, p – dichlorodiphenylchloroethane) is one of the best known pesticides. It is very effective against mosquito and lice. However, due to its harmful effects, its manufacture is banned;
Uses of carbon tetrachlorides (CCl4):
- It is used for manufacturing refrigerants and propellants for aerosol cans.
- It is used as feedstock in the synthesis of CFCs and other chemicals.
- It is used as a solvent in the manufacture of pharmaceutical products.
- CCl4 was once widely used as a cleaning fluid, a degreasing agent in industries, a spot remover homes, and a fire extinguisher.
Uses of iodoform (CHI3):
Iodoform was used as an antiseptic, but now it has been replaced by other formulations containing iodine-due to its objectionable smell. The antiseptic property of CHI3 is only due to the liberation of free iodine when it comes in contact with the skin.
![]()
Question 14.
Write the structure of the major organic product in each of the following reactions:

Answer:
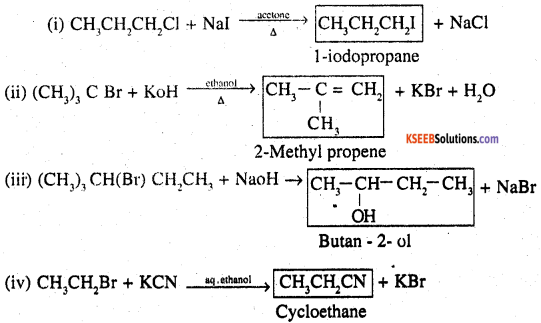
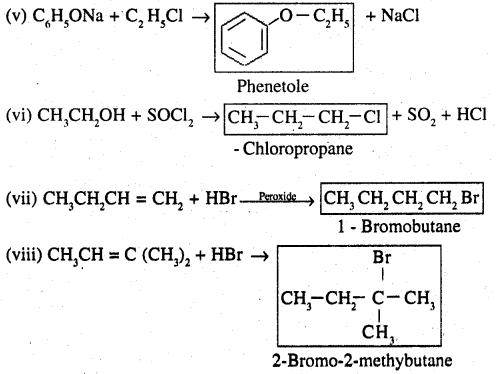
Question 15.
Write the mechanism of the following reaction:
![]()
Answer:
The given reaction is
![]()
The reaction is an SN2 reaction. In this reaction, CN– acts as the nucleophile and attacks the carbon atom to which Br is attacked. CN– is an ambident nucleophile and can attack through both C and N. In this case, it attacks through the C-atom.
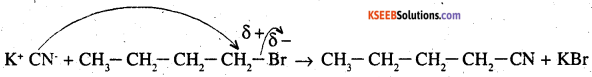
Question 16.
Arrange the compounds of each set in order of reactivity towards SN2 displacement:
1. 2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
2. l-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 3-Bromo-2-methylbutane
3. 1-Bromobutane, l-Bromo-2, 2-dimethylpropane, l-Bromo-2-methylbutane, l-Bromo-3-methylbutane.
Answer:
1.
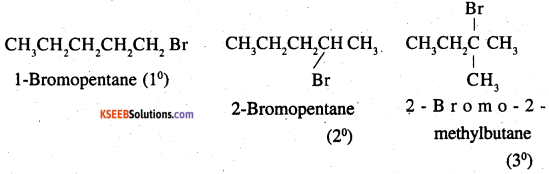
An SN2 reaction involves the approaching of the nucleophile to the carbon atom to which the leaving group is attached. When the nucleophile is sterically hindered, then the reactivity towards SN2 displacement decreases. Due to the presence of substituents, hindrance tp the approaching nucleophile increases in the following order.
1- Bromopentane < 2-bromopentane < 2 – Bromo-2-methyl butane
Hence, the increasing order of reactivity towards SN2 displacement is
2- Bromo – 2- methyl butane < 2-bromopentane < 1-Bromopentane
2.
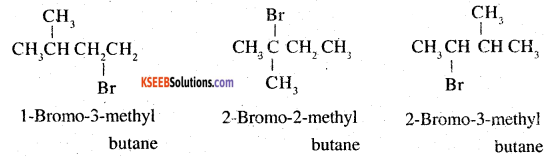
since steric hindrance in alkyl halides increases in the order of 1° <2° < 3°, the increasing order of reactivity towards SN2 displacement is 3° < 2° <1°.
Hence, the given set of compounds can be arranged in the increasing order of their reactivity towards
SN2 displacement as:
2-Bromo – 2- methyl butane < 3-bromo-2-methylbutane < 1-Bromo -3- methy lbutane.
3.
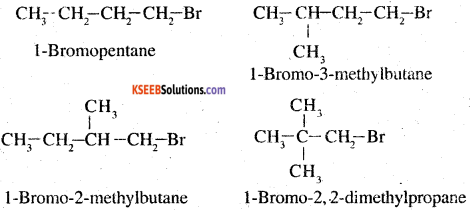
The steric hindrance to the nucleophile in the SN2 mechanism increases with a decrease in the distance of the substituents form the atom containing the leaving group. Further, the steric hindrance increases in the increases with increase in the number of substituents. Therefore, the increasing order of steric hindrances in the given compounds is as below:
1- Bromobutane < l-Bromo-3-methylbutane < 1 -Bromo-2-methylbutane < 1 -Bromo-2, 2- demethylbutane.
Hence, the increasing order of reactivity of the given compound towards SN2 mechanism is
l-Bromo-2, 2-dimethylbutane < 1 -Bromo-2-methyl butane < l-Bromo-3-methylbutane < 1-Bromobutane.
![]()
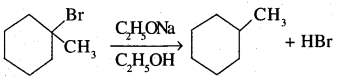
Question 17.
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH?
Answer:
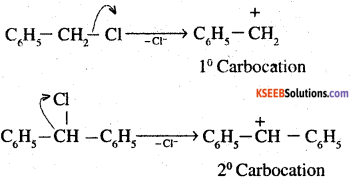
Question 18.
p-Dichlorobenzene has higher m.p. and solubility than those of o- and m-isomers. Discuss.
Answer:
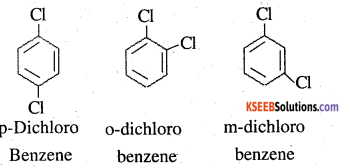
p-Dichlorobenzene is more symmetrical than o- and m- isomers. For this season, it fits more closely than o- and m-isomers in the crystal lattice. Therefore, more energy is required to break the crystal lattice of p-dichlorobenzene. As a result, p-dichlorobenzene has higher mp and lower solubility than o- and m- isomers.
Question 19.
How the following conversions can be carried out?
(i) Propene to propan-l-ol
(ii) Ethanol to but-l-yne
(iii) 1-Bromopropane to 2-bromopropane
(iv) Toluene to benzyl alcohol
(v) Benzene to 4-bromonitrobenzene
(vi) Benzyl alcohol to 2-phenylethanoic acid
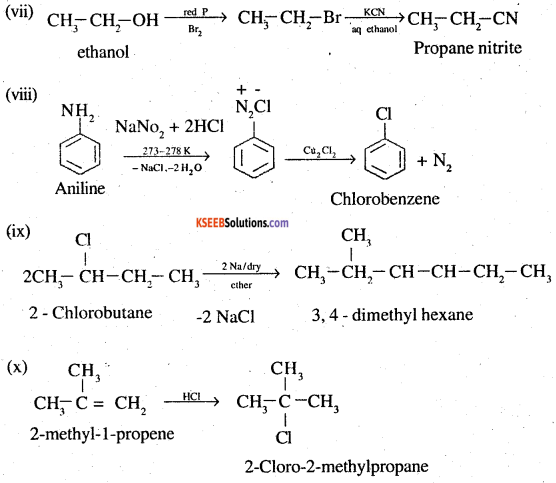
(vii) Ethanol to propanenitrile
(viii) Aniline to chlorobenzene
(ix) 2-ChIorobutane to 3, 4-dimethyIhexane
(x) 2-Methyl-l-propene to 2-chloro-2-methylpropane
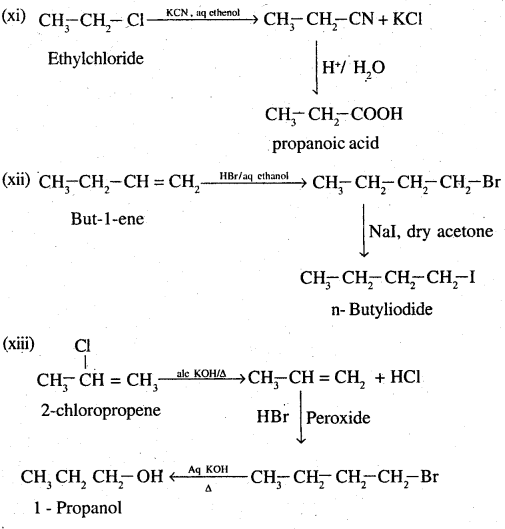
(xi) Ethyl chloride to propanoic acid
(xii) But-l-ene to n-butyliodide
(xiii) 2-ChIoropropane to 1-propanol
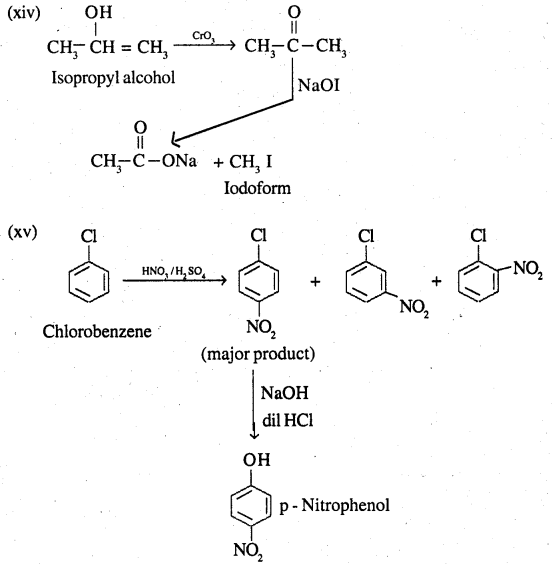
(xiv) Isopropyl alcohol to iodoform
(xv) Chlorobenzene to p-nitrophenol
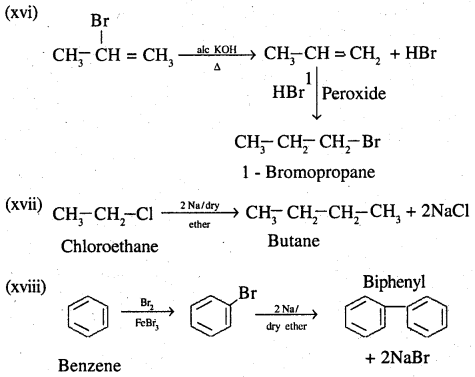
(xvi) 2-Bromopropane to 1-bromopropane
(xvii) Chloroethane to butane
(xviii) Benzene to diphenyl
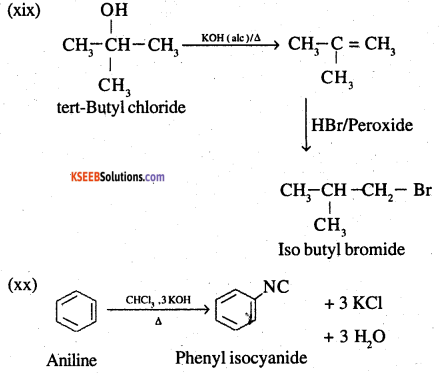
(xix) tert-Butyl bromide to isobutyl bromide
(xx) Aniline to phenylisocyanide
Answer:
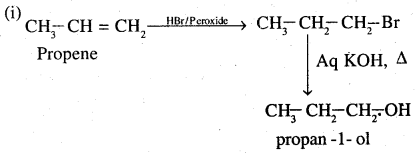
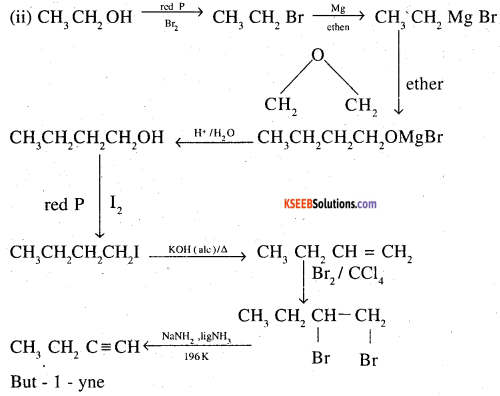
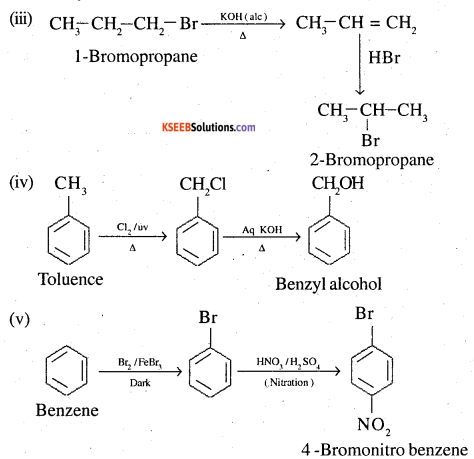
![]()
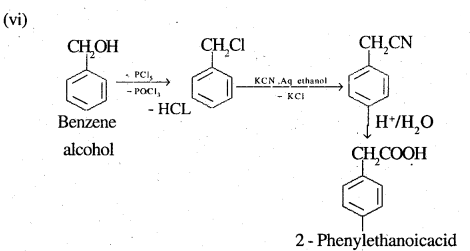
![]()
Question 20.
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Answer:
In an aqueous solution, KOH almost completely ionises to give OH– ions. OH– ions is a strong nucleophile, which leads the alkyl chloride to undergo a substitution reaction to form alcohol.
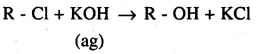
On the other hand, an alcoholic soln of KOH contains alkoxide (RO–) ion, which is a strong base. Thus, it can abstract a hydrogen from the β – carbon of the alkyl chloride and form an alkene by eliminating a molecule of HCl.
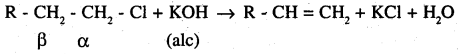
OH– ion is a much weaker base than RO– ion. Also, OH– ion is highly solvated in an aqueous solution and as a result the basic character of OH’ ion decreases. Therefore, it cannot abstract a hydrogen from β – carbon.
Question 21.
Primary alkyl halide C4H9Br (a) reacted with alcoholic KOH to give compound (b). Compound (b) is reacted with HBr to give (c) which is an isomer of (a). When (a) is reacted with sodium metal it gives compound (d), C8H18 which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (a) and write the equations for all the reactions.
Answer:
There are two primary alkyl halides having the formula C4H9Br. They one n-butyl bromide and isobutyl bromide
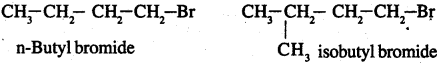
Since (a) reacts with Na to give a compound C8H18 which is different from the compound formed
when n – butyl bromide is reacted with Na, (a) has to be isobutyl bromide.
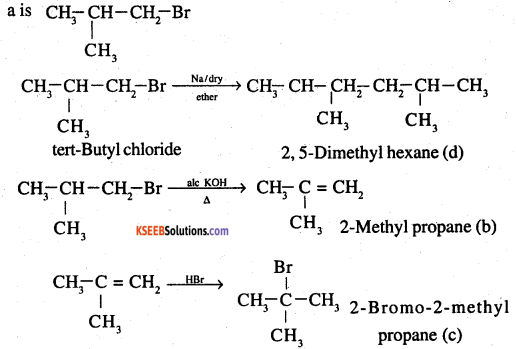
Question 22.
What happens when
(i) n-butyl chloride is treated with alcoholic KOH,
(ii) bromobenzene is treated with Mg in the presence of dry ether,
(iii) chlorobenzene is subjected to hydrolysis,
(iv) ethyl chloride is treated with aqueous KOH,
(v) methyl bromide is treated with sodium in the presence of dry ether,
(vi) methyl chloride is treated with KCN?
Answer:
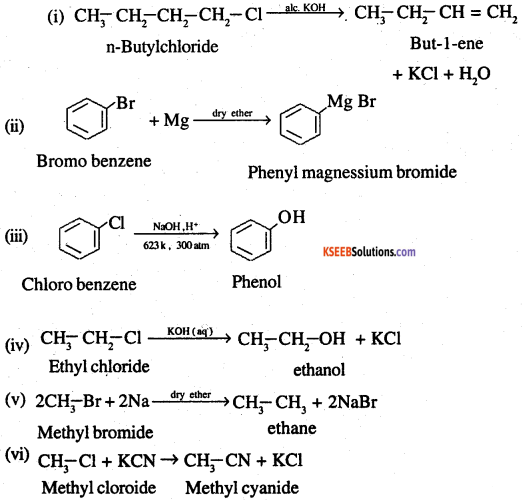
![]()