You can Download Chapter 8 The d-and f-Block Elements Questions and Answers, Notes, 2nd PUC Chemistry Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
Karnataka 2nd PUC Chemistry Question Bank Chapter 8 The d-and f-Block Elements
2nd PUC Chemistry The d-and f-Block Elements NCERT Textbook Questions and Answers
Question 1.
Write down the electronic configuration of:
(i) Cr3+
(ii) Pm3+
(iii) Cu+
(iv) Ce4+
(v) CO2+
(vi) Lu2+
(vii) Mn2+
(viii) Th4+
Answer:
Cr3+ : 1 s2 2s2 2p6 3s2 3p6 3d3 4s0 or [Ar] 3d3 4s0
Pm3+ : 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s6 5p6 6s0 5d0 4f4
Cu+ : 1s2 2s2 2p6 3s2 3p6 3d10 4s0 or [Ar] 3d10 4s0
Cr3+ : 1s2 2s2 2p6 3s2 3p6 3d3 4s0 or [Ar] 3d34s0
Ce4+ : 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s0 5d0 4f0 or [Xe] 4f4 5d0 6s0
Co2+ : [Ar] 3d7 4s0
Lu2+ : [Xe] 4f14 5d1 6s0
Mn2+ : [Ar] 3d5 4s0
Th4+ : [Rn] 5f0 6d2 7s2
Question 2.
Why are Mn2+ compounds more stable than Fe2+ towards oxidation to their +3 state?
Answer:

A half filled d orbital or a completely filled d orbital is more stable than any other state. Mn2+ already has a half filled stable state hence would not undergo oxidation to form Mn+3. On the other hand Fe2+ on oxidation to Fe3+ will have a half filled d orbitals which is more stable.
![]()
Question 3
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
Answer:
Except Sc, all other metals display +2 oxidation state. Also, on moving from Sc to Mn,’ the atomic number increases from 21 to 25; This means the number of e– in 3d orbital increases from 1 to 5
Sc2+ : d1 Ti2+ = d2 V2+ = d3 Cr2+ = d4
Mn2+ : d5
+ 2 oxidation state is attained by the loss of 2, 4s electrons by these metals since the number of d electrons in (+2) state also increases from Ti (+2) to Mn (+2), the stability of +2 state increases (as d – orbital is becoming more is half filled) M+2 has d5 electrons (that is half filled d shell, which is highly stable).
Question 4.
To what extent do the electronic configurations decide the stability of oxidation states in the first series of the transition elements? Illustrate your answer with examples.
Answer:
The electronic configurations play a very crucial role in deciding the stability of oxidation states. The presence or absence of complete or half filled d orbitals in that oxidation state decides its stability. Also the ability to give away e– decide the oxidation state which depends on the energy possessed by it and hence forth its electronic configuration,
For example
Sc shows +3 oxidation state only
[M] 3d1 4s2 +1, +2 very unstable due to presence of 1 d e- +3 oxidation state is stable with noble gas configuration.
Mn : [Ar] 3d5 4s2 has +2, +3, +4, +5, +6, +7 states but only +2 and +7 states are stable due to the half filled d orbital or completely empty d orbital.
similarly Ti = +2, +3, +4 with +4 most stable due to the same reason.
Question 5.
What may be the stable oxidation state of the transition element with the following d electron configurations in the ground state of their atoms : 3d3, 3d5,3d8 and 3d4?
Answer:

Question 6.
Name the oxometal anions of the first series of the transition metals in which the metal exhibits the oxidation state equal to its group number.
Answer:
Vanadate : VO3–
chromate : CrO42-
permanganate : MnO4–
Question 7.
What is lanthanoid contraction? What are – the consequences of lanthanoid contraction?
Answer:
The regular decrease in atomic radii due to the filling of 4f before 5d orbitals is called lanthanoid contraction. This is because of the imperfect shielding of one electron by another in the same set of orbitals. The shielding of one 4f electron by another is less than that of one d electron by another, and as the nuclear charge increases along the series, there is fairly regular decrease in the size of entire 4f orbitals. Consequences of lanthanoid contraction.
- Radii of third series are virtually same as those of the corresponding member of second series.
- Third series and second series have very similar physical and chemical properties much more than that expected by usual family relationships.
Question 8.
What are the characteristics of the transition elements and why are they called transition elements? Which of the d-block elements may not be regarded as the transition elements?
Answer:
Transition elements have incompletely filled d orbitals in its ground state or in any one of its oxidation states.
d-block elements are called transition elements and they lie between s and p block elements also all their properties are intermediate between s and p block elements.
Zn, Cd, Hg (elements of group 12) are not considered as transition metals because they have fully filled (d10) d orbitals configuration in its ground state as well as in their common oxidation state.
![]()
Question 9.
In what way is the electronic configuration of the transition elements different from that of the non transition elements?
Answer:
The general electronic configuration of a d-block elements is (n – 1) d1-10 ns1-2.
Transition elements in its ground state or excited state will have a configuration of (n – 1) d1-9 ns0-2 ie. partly filled d orbitals whereas non transition elements will have a configuration of (n – 1) d10 ns0.2 i.e completely filled d- orbitals.
Question 10.
What are the different oxidation states exhibited by the lanthanoids?
Answer:
The lanthanoids have an electronic configuration of 6s2 common with variable occupancy of 4f level. These elements have only one oxidation state(+3).
Question 11.
Explain giving reasons:
(i) Transition metals and many of their compounds show paramagnetic behaviour.
(ii) The enthalpies of atomisation of the transition metals are high.
(iii) The transition metals generally form coloured compounds. .
(iv) Transition metals and their many compounds act as good catalyst.
Answer:
(i) Paramagnetic arises due to the presence of unpaired electrons, each such electron has a magnetic moment associated with it due to its spin angular momentum. Transition metals have in its ground state or ionised state have number of unpaired d-electrons which gives it a paramagnetic behaviour.
(ii) Transition metals have very high interatomic metallic interaction due to the involvement of greater number of electrons from (n – l)d in addition to the ns electrons. Greater the number of valence electrons, stronger is the resultant bonding due to the greater overlapping of half filled orbitals. Hence, more amount of energy is required to break these metallic bond. Thus enthalpy of atomisation of transition metal is very high.
(iii) Colour of transition metal – compounds is due to the excitation of an electron from a lower energy d-orbital to a higher energy d orbital. The energy of excitation corresponds to the frequency of light absorbed and the colour observed corresponds to the complementary colour of the light absorbed (whose frequency lies on the visible region). The frequency of the light absorbed depends on the nature of ligand. Transition metals form coloured compounds due to presence of vacant d-orbitals for the d-d transition of e’ which causes the colour.
(iv) The catalytic activity of transition metals is ascribed to their’ability to adopt multiple oxidation states and to form complexes. Catalyst at a solid surface involve the formation of bonds between reactant molecules and atoms of the surface of the catalyst this has the effect of increasing the concentration of the reactants at the catalyst surface and also weakening of the bonds in the reacting molecules (activation energy is lowered. Transition metals have d and s orbitals to from these bonds.
Question 12.
What are interstitial compounds? Why are such compounds well known for transition metals?
Answer:
Interstitial compounds are those which are formed when small atoms like H, N or C are trapped inside the crystal lattices of metals. They are usually non-stoichiometric and are neither typically ionic nor covalent. Interstitial compounds are well known for transition compounds due to its closed crystalline structure with voids in them. The atomic size of transition metals is very large hence have large voids to occupy these small atoms.
Question 13.
How is the variability in oxidation states of transition metals different from that of the non transition metals? Illustrate with examples.
Answer:
The variability of oxidation states, a characteristics of transition elements arises out of incomplete filling of d orbitals in such a way that their oxidation states differ from each other by unity This is in contrast with the variability of oxidation states of non transition elements where oxidation states normally differ by a unit of two.
eg : Vanadium : V+2, V+3, V+4, V+5
Chromium : Cr+2, Cr+3, Cr+4, Cr+5, C+6
Nitrogen: +5, +3, +1, -1, -3.
Question 14.
Describe the preparation of potassium dichromate from iron chromite ore. What is the effect of increasing pH on a solution of potassium dichromate?
Answer:
Potassium Dichromates are generally prepared from chromates which in turn are obtained by the fusion of chromite ore (FeCr2O4) with sodium or potassium carbonate in free excess of air
4FeCr2O4 + 8Na2CO3 + 7O2 → 8Na2CrO4 + 2Fe2O3 + 8CO2
The yellow solution of Na2CrO4 is filtered and acidified with H2SO4 to give a solution from which orange sodium dichromate Na2Cr2O7 2H2O can be crystallised
2Na2CrO4 + 2H+→ Na2Cr2O7 + 2Na+ + H2O
sodium dichromate is more soluble than potassium dichromate. The latter is prepared by treating the solution of Na2Cr2O4 with KCl.
Na2Cr2O7 + 2KCl → K2Cr2O7 + 2NaCl (orange crystals)
(b) On increasing the PH of a solution of K2Cr2O7, it behaves like a strong oxidising agent and it oxidises iodide to iodine sulphides to sulphur, tin (II) to tin (IV) and Iron (II) salts to iron (III).
Question 15.
Describe the oxidising action of potassium dichromate and write the ionic equations for its reaction with:
(i) iodide (ii) iron(II) solution and (iii) H2S
Answer:
Potassium dichromate, in acidic medium acts as a strong oxidising agent by gaining 6e’ and forms Cr3+.
Cr2O72- + 14H+ + 6e– → 2Cr3+ + 7H2O ..(1)
(i) iodides
6I– → 3I2 + 6e– ..(2)
(1) + (2)
Cr2O72- + 14 H+ + 6I– → 2Cr3+ + 3I2 + 7H2O
(ii) iron (II) solution
6Fe2+ → 6Fe3+ + 6e– ..(3)
(1) + (3)
Cr2O72- +14 H++ 6Fe2+ → 2Cr3+ + 6Fe3+ + 7H2O
(iii) H2S
3H2S → 6H+ + 3S + 6e– ..(4)
(1) + (4)
Cr2O72- +14H+ + 3H2S → 6H+ + 3S + 2Cr3+ + 7H2O
![]()
Question 16.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with
(i) iron(II) ions
(ii) SO2 and
(iii) oxalic acid?
Write the ionic equations for the reactions.
Answer:
Potassium permangnate is prepared by fusion of MnO2 with an alkali metal hydroxide and an oxidising agent like KNO3. This produces the dark green k2MnO4 which disproportionates in a neutral or acidic solution to give permanganate.
2MnO2 + 4 KOH + O2 → 2K2MnO4 + 2H2O
3MnO42- + 4H+ → 2MnO4– + MnO2 + 2H2O
Commercially it is prepared by the alkaline oxidative fusion of MnO2 followed by the electrolytic oxidation of manganate (VI)
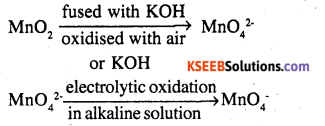
In laboralory a manganese (II) ion salt is oxidised by peroxidisulphate to permanganate.
2Mn2+ + 5S2O82- + 8H2O → 2MnO4– + 10SO42- + 16H+
(i) 5Fe2+ + MnO4– + 8H+ → Mn2+ + 4H2O + 5Fe37
(ii) 2MnO42+ 10SO2 + 5O2 + 4H2O → 2Mn2+ + 10SO42- + 8H+
(iii) 5C2O42- + 2MnO4– + 16H+ → 2Mn2+ + 8H2O + 10CO2
Question 17.
For M2+/M and M3+/M2+ systems the Eθ values for some metals are as follows:
Cr2+/Cr -0.9V
Mn2+/Mn -1.2V
Fe2+/Fe -0.4V
Cr3+/ Cr2+ -0.4 V
Mn3+/Mn2+ + 1.5 V
Fe3+/Fe2+ + 0.8 V
Use this data to comment upon:
(i) the stability of Fe3+ in acid solution as compared to that of Cr3+ or Mn3+ and
(ii) the ease with which iron can be oxidised as compared to a similar process for either chromium or manganese metal.
Answer:
(i) From the above data, we can infer that Fe3+ is less stable than Cr3+ but more stable than Mn3+. If reducing potential is less than oxidised state will be more stable. This is so because it is easier to add an e– to Fe3+ than Cr3+ but difficult to do so in case of Fe3+ than inMn3+.
(ii) Mn and Cr can be oxidised easily as compared to Fe because the reducing potential of Fe is less negative compared to Mn and Cr. Hence Fe is stable in its reduced form.
Question 18.
Predict which of the following will be coloured in aqueous solution?
Ti3+, V3+, Cu+, Sc3+, Mn2-, Fe3+ and CO2+.
Give reasons for each.
Answer:
Among the above mentioned, ions Ti3+, V3+ Mn2+, Fe3+, CO2+ will be coloured in its aqueous solution due to the ability of e’ to jump from a lower energy d orbital to a higher energy d orbital. In case of the ions Cu+, Sc3+ this d-d transition cannot take place either due to the absence of any e- in 3d orbital or due to complete filling of d orbital.
![]()
Question 19.
Compare the stability of +2 oxidation state for the elements of the first transition series.
Answer:
The relative stability of +2 oxidation state increases on moving from Sc to Zn. This is because on moving from top to bottom it becomes more and more difficult to remove the third e- from the d-orbital.
Question 20.
Compare the chemistry of actinoids with that of the lanthanoids with special reference to:
(i) electronic configuration
(ii) atomic and ionic sizes and
(iii) oxidation state
(iv) chemical reactivity.
Answer:
(i) Electronic configuration:
The general electronic configuration for lanthanoids is [Xe] 0-14 5d 0-1 6s2 and that of actinoids is [Rn] 5f 0-14 6d0-1 7s2. Unlike 4f orbitals, 5 f orbitals are not deeply buried and participate in bonding to a greater extent.
(ii) Oxidation states:
The principal oxidation states of lanthanoids is (+3). However sometimes we also encounter oxidation states of +2 and +4. This is because of extra stability of fully-filled and half filled orbitals. Actinoids exhibit a greater range of oxidation states. This is because the 5f, 6d and 7s levels are of comparable energies. Again, (+3) is the principal oxidation state for actinoids. Actinoids such as lanthanoids have more compounds in +3 state than in +4 state.
(iii) Atomic and ionic size Similar to lanthanoids, actinoids also exhibit actinoid contraction over all decrease in atomic and ionic radii. The contraction is greater due to the poor shielding effect of 5f orbitals. Hence there is an unexpected in the atomic and ionic sizes of actionoids. ‘
(iv) Chemical activity
In the lanthanoide series, the earlier members of the series are more reactive. They have reactivity that is comparable to Ca. With an increase in the atomic number, the lanthanides start behaving similar to Al. Actinoids, on the other hand, are highly reactive metals, especially when they are finely divided. When they are added to boiling water, they give a mixture of oxide and hydride. Actinoids combine with most of the non metals at moderate temperatures. Alkalies have no action on these actinoids. In case of acids, they are slightly affected by nitric acid (because of the -formation of a protective oxide layer).
Question 21.
How would you account for the following:
(i) Of the d4 species, Cr2+ is strongly reducing while manganese (III) is strongly oxidising.
(ii) Cobalt (II) is stable in aqueous solution but in the presence of complexing reagents it is easily oxidised.
(iii) The d1 configuration is very unstable in ions.
Answer:
(i) Cr2+ is strongly reducing in nature. It has a d4 configuration. While acting as a reducing agent, it gets oxidised to Cr3+ (electronic configuration d3). This d3 configuration can be written as t23 configuration, which is a more stable configuration. In the case of Mn3+ (d4) it Mn2+ (d5). This has an exactly half filled d – orbital and is highly stable.
(ii) Co (II) is stable in aqueous solutions however, in the presence of strong field complexing is agents, it is oxidised to Co (II). Although the 3rd ionisation energy for Co is high, but the higher amount of crystal field stabilisation field ligands overcomes this ionisation energy.
(iii) The ions in d configuration tend to lose one more electrons to get into stable d0 configuration. Also, the hydration or lattice energy is more than sufficient to remove the only electron present in the d-orbital of these ions. Therefore, they act as reducing agents.
![]()
Question 22.
What is meant by ‘disproportionation’? Give two examples of disproportionation reaction in aqueous solution.
Answer:
It is found that sometimes a relatively less stable oxidation state undergoes an oxidation reduction reaction in which it is simultaneously oxidised and reduced. This is called disproportion.
For example,
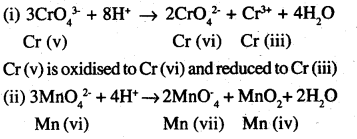
Mn (vi) is oxidised to Mn (vii) and reduced to Mnfiv).
Question 23.
Which metal in the first series of . transition metals exhibits +1 oxidation state most frequently and why?
Answer:
In the fast transition series, Cu exhibits +1 oxidation state very frequently. It is because Cu (+1) has an electronic configuration of [Ar] 3d10. The completely filled d – orbital makes it highly stable.
Question 24.
Calculate the number of unpaired electrons in the following gaseous ions: Mn3+, Cr3+, V3+ and Ti3+. Which one of these is the most stable in aqueous solution?
Answer:

Cr3+ is the most stable in aqueous solution owing to a t23 g configuration.
Question 25.
Give examples and suggest reasons for the following features of the transition metal chemistry:
(i) The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
(ii) A transition metal exhibits highest oxidation state in oxides and fluorides.
(iii) The highest oxidation state is exhibited in oxoanibns of a metal.
Answer:
(i) In case of a lower oxide of a transition metal, the metal atom has a low oxidation state. This means that some of the valence electrons of the metal atoms are not involved in bonding. As a result, it can donate electrons and behave as a base.
On the other hand, in the case of a higher oxide of a transition metal, the metal atom has a high oxidation state. This means that the valence electrons are involved in bonding. As a result, it can accept electrons and behave as an acid. For example, MnIIO is basic and MnVIIO is acidic.
(ii) Oxygen and fluorine act as strong oxidising agents because of their high electronegativities and small sizes. Hence, they bring out the highest oxidation states from the transition metals. In other words, a transition metal exhibits oxidation states in oxides and fluorides. For example, in OsF6 and V2O5, the oxidation states of Os and V are + 6 and respectively.
(iii) Oxygen is a strong oxidising agent due to its high electronegativity and small size so, oxoanions of a metal have the highest oxidation state. For example, in MnO4–, the oxidation state of Mn is +7.
![]()
Question 26.
Indicate the steps in the preparation of:
(i) K2Cr2O7 from chromite ore.
(ii) KMnO4 from pyrolusite ore.
Answer:
(i) Potassium dichromate : (K2Cr2O7) is prepared from chromite ore (FeCl2O4) in the following steps.
step (1) Preparation of sodium chromate
4Fe Cr2O4 + 16 NaOH + 7O2 → 8Na2CrO4 + 2Fe2O3 + 8H2O
step (2) Conversion of sodium chromate into sodium dichromate
2Na2CrO4 + con H2SO4 → Na2Cr2O7 + Na2SO4 + H2O
step (3) Conversion of sodium dichromate to potassium dichromate
Na2Cr2O7 + 2KCl → 2Nacl + k2Cr2O7
potassium chloride being less soluble than sodium chloride is obtained in the form of orange coloured crystals and can be removed by filteration.
The dichromate ion (Cr2O72- ) exists in equilibrium with chromate (CrO42- ) ion at pH.
4. However, by changing the pH, they can be interconverted.
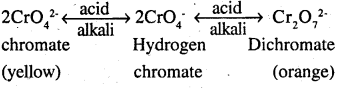
(ii) Potassium permanganate : (KMnO4) can be prepared from pyrolusite (MnO2). The ore is fused with KOH in the presence of either atmospheric oxygen or an oxidising agent, such as KNO3 or KClO4 to give K2MnO4

The green mass can be extracted with water and then oxidised either electrolytically or by passing chlorine / ozone into the solution, electrolytic oxidation
K4MnO4 2K++ ↔️ MnO42-
H2O ↔️ H+ + OH–
at anode manganate ions are oxidized to permanganate ions
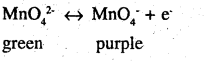
oxidation by chlorine
2K2MnO4 + Cl2 → 2KMnO4 + 2KCl
2MnO42- + Cl2 → 2 MnO4 + 2Cl–
oxidation by ozone
2K2MnO4 + O3 + H2O → 2KMnO4 + 2KOH + O2
2MnO42- + O3 + H2O → 2MnO2 + 2OH + O2
![]()
Question 27.
What are alloys? Name an important alloy which contains some of the lanthanoid metals. Mention its uses.
Answer:
An alloy is a solid or solution of two or more elements in a metallic matrix. It can either be a partial solid solution or a complete solid solution. Alloys are usually found to posses different physical properties than those of the component elements.
An important alloy of lanthanoids is Mischmetal It contains lanthanoids (4-95%), iron (5%) and traces of S, C, Si, Ca and Al.
Uses
- Mischmetal is used in cigarettes and gas lighters
- It is used in flame throwing tanks
- It is used in flame bullets and shills.
Question 28.
What are inner transition elements? Decide which of the following atomic numbers are the atomic numbers of the inner transition elements : 29, 59, 74, 95, 102, 104.
Answer:
Inner transition metals are those elements in which the last electron enters the f-orbital. The elements in which the 4f and the 5f orbitals are progressively filled are called f-block elements. Among the given atomic numbers the atomic numbers of the inner transition u elements are 59, 95 and 102.
Question 29.
The chemistry of the actinoid elements is not so smooth as that of the lanthanoids. Justify this statement by giving some examples from the oxidation state of these elements.
Answer:
Lanthanoids primarily show three oxidation states, +3 states is the most common. Lanthanoids display a limited number of oxidation states because the difference between the energies of 4f, 5d and 6s orbitals is quite large. On the other hand, the energy difference between the 5f, 6d and 7s orbitals is very less. Hence, actinoids display a large number of oxidation states. For example uranium and plutonium display +3, +4, +5 and +6 oxidation states while neptunium display +3, +4, +5 and +7. The most common oxidation state in case of actinoids is also +3.
Question 30
Which is the last element in the series of the actinoids? Write the electronic configuration of this element. Comment on the possible oxidation state of this element.
Answer:
The last element in the actinoid series is Lawrencium. Lr. Its atomic number is 103 and its electronic configuration is [Rn] 5f14 6d17s2. The most common oxidation state displayed by it is +3, because after losing 3 electrons it attains stable f14 configuration.
Question 31
Use Hund’s rule to derive the electronic configuration of Ce3+ ion, and calculate its magnetic moment on the basis of ‘spin- only’ formula.
Answer:
Ce : 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 sp6 4f1 5d1 6s2
Ce : [Xe] 4f1 5d1 6s2
Ce3+: {Xe} 4f1
ForCe3+:
Number of unpaire electrons = n = 1
\(\mu=\sqrt{\mathrm{n}(\mathrm{n}+2)}\)
= √8
= 2√2
= 2.828 BM.
Question 32.
Name the members of the lanthanoid series which exhibit +4 oxidation states and those which exhibit +2 oxidation states. Try to correlate this type of behaviour with the electronic configurations of these elements.
Answer:
The lanthanoids that exhibit +2 and +4 states are shown in the given table. The atomic number of the elements are given in the parenthesis.

Ce after forming Ce4+ attains a stable configuration of [Xe]
Tb after forming Tb4+ attains a stable configuration of [Xe] 4f7
Eu after forming Eu2+ attains a stable configuration of [Xe] 4f7
Yb after forming Yb2+ attains a stable configuration of [Xe] 4f14.
Question 33.
Compare the chemistry of the actinoids with that of lanthanoids with reference to:
(i) electrpnic configuration
(ii) oxidation states and
(iii) chemical reactivity.
Answer:
(i) Electronic configuration:
The general electronic configuration for lanthanoids is [Xe] 0-14 5d 0-1 6s2 and that of actinoids is [Rn] 5f 0-14 6d0-1 7s2. Unlike 4f orbitals, 5 f orbitals are not deeply buried and participate in bonding to a greater extent.
(ii) Oxidation states:
The principal oxidation states of lanthanoids is (+3). However sometimes we also encounter oxidation states of +2 and +4. This is because of extra stability of fully-filled and half filled orbitals. Actinoids exhibit a greater range of oxidation states. This is because the 5f, 6d and 7s levels are of comparable energies. Again, (+3) is the principal oxidation state for actinoids. Actinoids such as lanthanoids have more compounds in +3 state than in +4 state.
(iii) Atomic and ionic size Similar to lanthanoids, actinoids also exhibit actinoid contraction over all decrease in atomic and ionic radii. The contraction is greater due to the poor shielding effect of 5f orbitals. Hence there is an unexpected in the atomic and ionic sizes of actionoids. ‘
(iv) Chemical reactivity
In the lanthanoide series, the earlier members of the series are more reactive. They have reactivity that is comparable to Ca. With an increase in the atomic number, the lanthanides start behaving similar to Al. Actinoids, on the other hand, are highly reactive metals, especially when they are finely divided. When they are added to boiling water, they give a mixture of oxide and hydride. Actinoids combine with most of the non metals at moderate temperatures. Alkalies have no action on these actinoids. In case of acids, they are slightly affected by nitric acid (because of the -formation of a protective oxide layer).
![]()
Question 34.
Write the electronic configurations of the elements with the atomic numbers, 61, 91, 101, and 109.
Answer:
61 : [Xe]54 4f5 5d0 6s2
91 : [Rn]86 5f2 6d1 7s2
101 : [Rn]86 5f13 5d0 7s2
109: [Rn]86 5f14 6d7 7s2 .
Question 35
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following points:
(i) electronic configurations
(ii) oxidation states
(iii) ionisation enthalpies and
(iv) atomic sizes.
Answer:
(i) In the 1st , 2nd and 3rd series of the transition metals the 3d, 4d, 5d orbitals are respectively filled.
We know that elements in the same vertical column generally have similar electronic configurations.
In the first transition series, two elements show unusual electronic configurations.
Cr (24) : 3d5 4s1
Cu (29) :3d10 4s1
similarly, there are exception in 2nd transition series.
These are
Mo (42) : 4d5 5s1
Tc (43) : 4d6 5s1
Ru (44) : 4d7 5s1
Rh (45) : 4d8 5s1
Pd (46) : 4d10 5s1
Ag (47) : 4d10 5s1.
There are exceptions in-3rd transition series also.
These are
W (74) : 5d4 6s2
Pt (78) : 5d9 6s1
Au (79) :5d10 6s1
As a result of these exceptions it happens many times that the electronic configuration of the elements presents in the same group are dissimilar.
(ii) In each of the three transition series the number of oxidation states shown by the elements is the maximum iri the middle and the minimum at the extreme ends. However, +2 and +3 oxidation states are quite stable for all elements present in the first transition series. All metals present in the first transition series form stable compounds in the +2 and +3 oxidation states. The stabilities of the +2 and +3 oxidation states decreases in the second and the third transition series, where in higher oxidation states are more important.
For example, [FeII (CN)6]4- [COIII (NH3)6]3+ , [Ti(H2O)6]3+ air stable complexes, but no such complexes are known for the second and third transition series such as Mo, W, Rh, In. They form complexes in which their oxidation states are high. For example WCl6, ReF7, RuO4 etc.
(iii) In each of the three transition series, the first ionisation enthalpy increases from left to right. However, there are some enceptions. The first ionisation enthalpies of the third transition series are higher than those of the first and second transition series. This occurs due to poor shielding effect of 4f electrons in the third transition series.
Certain elements in the second transition series have higher first ionisation enthalpies than elements corresponding to the same vertical column in the first transition series. There are also elements in the 2nd transition series whose first ionisation enthalpies are lower than those of the elements corresponding to the same vertical column in the 1st transition series.
(iv) Atomic sizes generally decreases from left to right across a period. Now, among the three transition series, atomic sizes of the elements in the second transition series are greater than those of the elements corresponding to the same vertical column in the first transition series. However, the atomic sizes of the elements in the third transition series are virtually the same as those of the corresponding members in the second transition series this is due to lanthanoid contraction.
Question 36.
Write down the number of 3d electrons in each of the following ions:
Ti2+ , V2+ , Cr3+ , Mn2+ , Fe2+ , Fe3+ , CO2+ , Ni2+ and Cu2+ .
Indicate how would you expect the five 5d orbitals to be occupied for these hydrated ions (octahedral).
Answer:

Question 37.
Comment on the statement that elements of the first transition series possess many properties different from those, of heavier transition elements.
Answer:
The properties of the elements of the first transition series differ from those of the heavier transition elements in many ways.
(i) The atomic sizes of the elements of the first transition series are smaller than those of the heavier elements of 2nd and 3rd transition series) However, the atomic sizes of the elements in ‘the third transition series are virtually the same as those of the corresponding members in the second transition series this is due to lanthanoid configuration.
(ii) +2 and +3 oxidation states are more common for elements in the first transition series, while higher oxidation states are more common for the heavier elements.
(iii) The enthalpies of atomisation of the elements’in the first transition series are lower than those of the corresponding elements in the second and third transition series.
(iv) The meeting and boiling points of the first transition series are lower than those of the heavier transition elements. This is because of the occurrence of strong metallic bonding (M – M bonding)
(v) The dements of the first transition series from low-spin or high-spin complexes depending upon the strength of the ligand field.
However, the heavier transition elements form only low-spin complexes, irrespective of the strength of the ligand field.
![]()
Question 38.
What can be inferred from the magnetic moment values of the following complex species ?

Answer:
Magnetic moment (μ) is givenn as

For transition metals, the magnetic moment is calculated from the spin only formula Therefore
\(\sqrt{n(n+2)}\) = 2.2
we can see from above calculation that the given value is closest to n – 1. Also, in this complex,. Mn is in the +2 oxidation state. This means that Mn has 5 electrons in the d orbital. Hence, we can say that CN is a strong ligand that causes the pairing of electrons.
(ii) [Fe (H2O)6]2+
We. can see from the above calculation that µ= 5.3 is closest to the value when n = 4. Also, in this complex, Fe is in the +2 oxidation state. This means ihat Fe has 6 electrons in the d orbital
Hence, we can say that H2O is a weak field ligand and does not cause the pairing of e– s.
(iii) K2 [Mncl4]
we can see that |X= 5.9 is closest to the value when n = 5. Also, in this computer, Mn is in the +2 oxidation state. This means that Mn has 5 electrons in the d-orbital. Hence we can say that Cl- is a weak ligand and does not cause the pairing of electrons.
2nd PUC Chemistry The d-and f-Block Elements Additional Questions and Answers
Question 1.
Some d-block elements are given below.
(Cr, Mn, Fe, Co, Ni)
(a) Identify the element which shows maximum paramagnetic behaviour.
(b) Give reason for the highest paramagnetic property of that element.
(c) When two electrons are lost from each atom, is there any change in magnetic properties of each ? Explain ?
Answer:
(a) Cr
(b) Cr- 4s1 3d5
Mn – 4s2 3d5 .
Fe – 4s2 3d6
Co – 4s2 3d7
Ni – 4s2 3d8
Since there are six unpaired electrons in Cr, it has the maximum paramagnetic behaviour.
(c) Cr2+ →3d4
Mn2+ → 3d5
Fe2+ → 3d6
CO2+ → 3d7
Ni2+ → 3d8
when two electrons are lost, Mn2+ contains maximum number of unpaired electrons ie., Mn2+ is the ion with maximum paramagnetic behaviour. Cr2+ and Fe2+ show similar paramagnetism and so on.
Question 2.
How do atomic radii of transition elements in a given series vary with increase in atomic number ? What is your observation on atomic radii of 4d and 5d series of transition elements ? Discuss the phenomenon involved, if any
Answer:
The 4d and 5d series of transition elements in each group have the same atomic radii. This is due to lanthanide contraction.
The steady decrease in the size of the atoms or ions from lanthanum to lutetium is known as lanthanide contraction.
As the atomic number increases in the lanthanide series, the new electron goes to the inner 4f orbitals. The shielding effect of a 4f electron is poor. As a result, the attraction of the outer electrons by the nucleus increases and this tends to a decrease in the size.
Question 3.
(a) What is blue Vitriol ? How is it useful in detecting the presence of water vapour ?
(b) When a magnetic field is applied, certain substances are attracted by the applied field. Those are called paramagnetic substances. Which is more paramagnetic Mn2+ or Cu2+ ? Why ?
Answer:
(a) CuSO4 5H2O is known as blue vitriol. Anhydrous copper sulphate is used for detecting the presence of water vapour. Water vapour converts white anhydrous copper sulphate to blue hydrated copper sulphate.
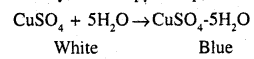
(b) As the number of unpaired electrons increases, paramagnetism increases.
Mn → [Ar] 4s2 3d5 and Mn2+ → 3d5
Cu → [Ar] 4s1 3d10 and Cu2+ →3d9
Mn2+ contains five unpaired d-electrons and
Cu2+ contains only unpaired electron.
∴ Mn2+ is more paramagnetic than 2+.
![]()
Question 4.
(a) What do you observe when a dilute solution of KMnO4 is added in drops to a hot solution of dilute H2SO4 ? Is there any difference in observation when dilute KMnO4 is added to a mixture of hot dilute H2SO4 and oxalic acid ?
(b) In the periodic table the atomic size increases down a group due to increase in number of shells. But 40Zn and 72HF in the 5th and 6th period respectively have almost identical radii explain ?
Answer:
(a) When dilute KMnO4 is added to a mixture of hot dilute H2SO4 and oxalic acid, brisk effervescence is formed due to the formation of CO2.
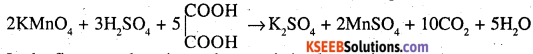
In the first case there is no characteristic observation
(b) The 4d and 5d series of transition elements in each group have the same atomic radii. This is due to lanthanide contraction.
The steady decrease in the size of the atoms or ions from lanthanum to lutetium is known as lanthanide contraction.
As the atomic number increases in the lanthanide series, the new electron goes to the inner 4f orbitals. The shielding effect of a 4f electron is poor. As a result, the attraction of the outer electrons by the nucleus increases and this tends to a decrease in the size.
Question 5.
Mn exhibits large number of oxidation states in its compounds. Which is the highest oxidation state ?
Name an industrially important compound you have studied in which Mn is in its highest oxidation state. How is that compound manufactured ?
Answer:
In KMnO4, Mn exhibits the highest oxidation state of +7.
Oxygen and fluorine act as strong oxidising agents because of their high electronegativities and small sizes. Hence, they bring out the highest oxidation states from the transition metals. In other words, a transition metal exhibits oxidation states in oxides and fluorides. For example, in OsF6 and V2O5, the oxidation states of Os and V are + 6 and respectively.
Question 6.
Assign reasons for the followimg.
(i) The enthalpies of atomisation of transition elements are high.
(ii) The transition metals and many of their compounds act as good catalyst.
(iii) From element to element the actinoid contraction is greater than the lanthanoid contraction
(iv) The E° value for the Mn3+/Mn2+ couple is much more positive than for Cr3+/Cr2+
(v) Scandium (Z = 21) does not exhibit variable oxidation states and Let it is regarded as transition element.
Answer:
(i) The transition elements exhibit high enthalpies of atomisation because they have large number of unpaired electron in their atoms. Due to this they have stronger inter atomic interaction and hence stronger bonding between atoms.
(ii) Many transition metals and their compounds acts as catalyst the catalytic activity is due to their ability to exhibit multiple oxidation states, eg. V2O5 in contact process and finely divided iron in Haber process.
(iii) The actinoid contraction is similar to lanthanoid contraction. This contraction is due to poor shielding by 5f electrons in the actioids than that by 4f electrons in the lanthanoids.
(iv) Much larger third ionisation energy of Mn (where the required change is Cl5 and d4) is mainly responsible for this. This also explains that +3 state of Mn is of little importance.
(v) Scandium (Z = 21) has incompletely filled 3d orbitals in the ground state. (3d) Hence, it is considered as a transition element.
Question 7.
(a) What may be the possible oxidation states of the transition metals with the following d
electronic configurations for the ground state of their atoms 3d3 4s2, 3d5 4s2 and 3d6 4s2. Indicate relative stability of oxidation states in each case.
(b) Write steps involved in the preparation of
(i) Na2CrO4 from chromite ore and
(ii) K2MnO4 from pyrolusite ore.
Answer:
(a) The most stable oxidation state for 3d3 4s2 = +5
The most stable oxidation state for 3d5 4s2 = +7, +2
The most stable oxidation state for 3d6 4s2 = +2
In a transition series, the oxidation states which ‘ lead to exactly half-filled or completely filled d- orbitals are more stable. It shows that oxidation states +2 to +7 is most stable.
(b) i. The powdered chromite ore is heated with sodium carbonate in presence of air to get yellow coloured sodium chromite.
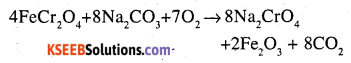
ii. Finally powdered pyrolusite is fused with KOH in the presence of air to give green coloured potassium manganate
2MnO2 + 4 KOH +O2 → 2K2MnO4 + 2H2O.
Question 8.
Write complete chemical equations for
(i) Oxidation of Fe2+ by Cr2O72- in acid medium
(ii) Oxidation S2O32- by MnO4– in neutral aqueous medium
Answer:
(i) Acidified potassium dichromate oxidises iron (II) salt to iron (III)
Cr2O72- + 14H + 6Fe2+ → 2Cr3+ + 6Fe3+ + 7H2O
(ii) In natural aqueous medium, thiosulphate
8MnO4–+ 3s2O32- + H2O → 8MnO2+ 65O4 + 2OH–
Question 9.
How would you account for the following situations
(i) The transition metals generally form coloured compounds.
(ii) With 3d4 configuration, Cr2+ acts as a reducing agent but Mn3+ acts as an oxidising agent (Atomic masses, Cr = 24, Mn = 25)
(iii) The actinoids exhibits a larger number of oxidation states than the corresponding lanthanoids.
Answer:
(i) Colour of compounds of transition elements depends upon the unpaired electrons present in d orbitals of transition elements. If d orbitals are completely vacant as in Sc3+, Ti4+ or completely filled as in Cu2+, Zn2+, the compounds will be colourless. But if any unpaired electron is present in d-orbitals, the compound will be coloured due to d → d transition. The unpaired electron is excited from one energy level to another energy level within the same d subshell. For this purpose (d → d transition), the energy is absorbed from visible region of radiation and reflected light will decide the colour of the compound.
(ii) Cr2+ is strongly reducing in nature. It has a d4 configuration. While acting as a reducing agent, it gets oxidised to Cr3+ (electronic configuration d3). This d3 configuration can be written as t23 configuration, which is a more stable configuration. In the case of Mn3+ (d4) it Mn2+ (d5). This has an exactly half filled d – orbital and is highly stable.
(iii) Actinoids show a large number of oxidation states because of small energy gap between 5f, 6d and 7s subshells.
![]()